With updates coming for USP-797 and USP-800, pharmaceutical labs need to make some fundamental changes to stay in regulation. Updates to these pharmaceutical regulations are expected to go nationwide in December, so let’s start by taking a more in-depth look at what changes you can expect to see.
Significant changes will be in the following areas:
- Calibrations, certifications, and qualifications of equipment and classified areas
- Requirements for accreditation of classified cleanroom areas
- Testing performed as part of the facility certification
- Scenarios requiring Investigations and Corrective Actions
- Vented biological safety cabinet requirements
- Containment secondary engineering control (C-SEC)
- Control requirements


While each facility will require different updates to stay compliant, new, or recently renovated pharmacies should be especially careful. If your lab was designed to meet minimum standards, you might have a little more work to do than other labs.
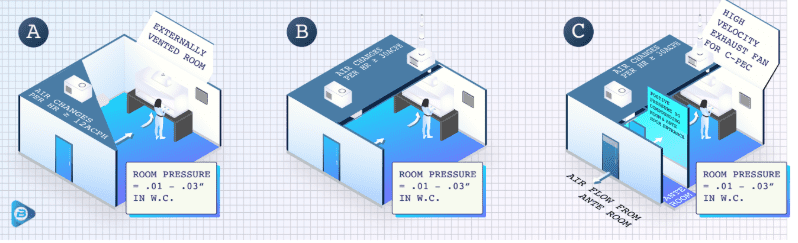
Compliance under these regulations is critical for the safety of technicians and patients. The graphics below show setups for non-sterile, sterile, and containment hazardous drug compounding pharmacies. Each of these setups requires different secondary controls to control the lab environment accurately. Check them out to find out what your pharmacy needs to update:
- Non-Sterile HD Room
- Sterile HR Room
- Containment Segregated Compounding Area

Background on USP-797 and USP-800
Pharmaceutical Regulation USP-797:
USP-797 was designed to protect both patients and pharmacists. It classified sterile compounding into three risk groups: Low, Medium, and High. This regulation applies to all pharmacies that are producing a compound, sterile preparations, or CSPs. The chapter is specifically designed to prevent harm and fatality form microbial contamination, excessive bacterial endotoxins, and abundant content errors of certain ingredients in CSPs.
Pharmaceutical Regulation USP-800:
USP-800 expands on USP-797 and is designed to describe the requirements and responsibilities for handling hazardous drugs, facility and engineering controls, procedures for deactivating, decontamination, and cleaning; and spill control and documentation.
USPs are Making a Difference
Both USP-797 and USP-800 pharmaceutical regulations are helping healthcare practitioners by making compounding safer. All together, USP General Chapters are standards to assist practitioners in creating high-quality medicine and practices. They ensure patients are getting the right dosage, quality, and contaminant-free medication every time they go into their pharmacy.
“From the time a medication is compounded until the moment the patient takes it, we want to help ensure the patient benefit and reduce risks,” said Ronald T. Piervincenzi, Ph.D., chief executive officer, USP. “The updated USP standards will help ensure quality preparations for patients who rely on compounded medicines while minimizing the risk of exposure to hazardous drugs for healthcare personnel.”
Pharmacy #regulations are changing. Is your lab ready? Let BalCon lead you through the changes to help you get ready! Click To TweetAt BalCon, we are available for consultation to help you through all stages, from design to occupancy of your cleanroom suites. We’ll work with you to:
- Avoid costly changes and delays during your project.
- Assist with cleanroom monitoring needs such as room pressurization, temperature, and humidity.
- Discuss certification strategies to minimize downtime.
- Discuss documentation and report reviews to help you better understand your cleanroom reports.
- Assist with GAP Analysis to evaluate your pharmacy for compliance.
Not sure where to start? Contact BalCon today and let us get your lab suited for these new pharmaceutical regulations.



