Deaerators are a key component to many commercial and industrial boiler systems that require regular maintenance to ensure long and efficient equipment life.

Deaerator tanks serve 3 primary purposes.
1. They store and supply consistent water to the boiler.
2.They bring the temperature of the water up before it supplies it to the boiler. They do this by mixing fresh make-up water with steam. This helps to prevent thermal shock as well as improve overall boiler room efficiency.
3. They drive undissolved oxygen and other gasses out of the water before it enters the boiler.
The third item is very important because oxygen and other gasses can wreak havoc inside a boiler. Oxygen and CO2, even in trace amounts as low as 5 ppm (parts per million), will cause major corrosion damage over time. Dissolved oxygen will react with carbon steel throughout the feed water piping, economizers, and boiler, causing oxygen pitting and eventually leaks.
In most applications you are looking for 5-7 psi of pressure and a temperature of 225 to 227 degrees. This will ensure the DA Tank can remove these gases and get the water up to temperature before supplying it to the boiler.
HOW A DEAERATOR WORKS

While there are many types of deaerators, they generally behave the same. Feedwater enters the DA through an inlet water connection. The steam in the tank then interacts with the make-up water which raises the water’s temperature. This breaks oxygen and other undissolved gasses up and allows them to escape through a vent located at the top of the DA tank.
The water then passes through a baffled scrubber section of the tank and is scrubbed with oxygen free steam. Steam then goes through a spray valve which turns the steam into a fine mist. The water is then ready for use by the boiler as the gases released vent to the atmosphere.
WHY DEAERATE?

As mentioned above, a deaerator helps prevent corrosion. Corrosion occurs when oxygen in the water reacts with iron in the system. If you do nothing to remove the oxygen, the iron in the system will! The metal will react chemically & disintegrate, forming rust.
The chemical process starts when iron interacts with water to form Ferrous Hydroxide.
Fe + 2H2O = Fe(OH)2 + 2H+ Iron + Water = Ferrous Hydroxide + Hydrogen
(the top layer of Ferrous Hydroxide protects the remaining iron)
If dissolved oxygen is present, it combines with the ferrous hydroxide to form an insoluble compound, ferric hydroxide, which is rust.
4Fe (OH)2 + O2 + H2O = 4Fe(OH)3
Ferrous Hydroxide + Oxygen = Ferric Hydroxide (Rust)
If the system continuously cycles dissolved oxygen, the ferrous hydroxide will be continuously removed from the system until the metal has been totally dissolved.
WHAT NEEDS TO HAPPEN FOR DEAERATION TO OCCUR?
1. Temperature – Water cannot absorb gasses when it is heated to saturation temperature. Water also only releases gasses when it reaches saturation temperature.
2. Turbulence – This helps to break the surface tension of water and aids in the release of gases.
3. Time – You need to have enough time for the release of gases to occur.
4. Venting – The gases that are released in the steps above need to be able to escape.
5. Steady State Conditions – without steady state conditions, the deaeration process can not occur!
A constant flow of water through spray nozzles or trays insures adequate turbulence.
Stead flow of feed water, with only gradual temperature changes, allows pressure controllers to satisfy demands for steam.
Steady flows of feed water also assures that the deaerator will operate with the capacity for which it was designed.
WHEN DO YOU NEED A DEAERATOR INSTEAD OF A BOILER FEED SYSTEM?
1. Higher levels of make-up provide more Oxygen which leads to:
Increased chemical use
Greater blowdown
Higher energy losses
Increased make-up
2. Higher operating pressures leads to increased CO2 production:
Lower pH condensate
Higher levels of non-condensable gases in heat exchangers
3. Higher operating temperatures in the boiler increase the need for feedwater heat.
WHEN SELECTING A DEAERATION SYSTEM
The Deaeration System requires steady state conditions to operate
Deaerators require constant steam pressure to operate
The Deaeration System must isolate the boiler from the steam distribution system
The Deaerator must operate under all load conditions which the system experiences
Types of Deaerators And There Operation
There are two main types of deaerators: spray type and tray type. (It is important to note that there are other types of DA’s, but we are focusing on the two most common)
Spray Type
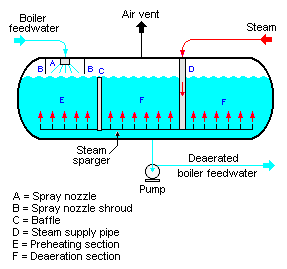
According to STS Canada; Spray-Type Deaerators serve as both the deaeration section and the boiler feed water storage tank.
The typical spray-type deaerator is a horizontal vessel which has a preheating section and a deaeration section. A baffle separates the two sections. Low-pressure steam enters the vessel through a sparger in the bottom of the vessel.
The purpose of the feed water spray nozzle and the preheat section is to heat the boiler feed water to its saturation temperature. This facilitates stripping out the dissolved gases in the following deaeration section.
The preheated feed water then flows into the deaeration section, where it is deaerated by the steam rising from the sparger system.
Gases stripped out of the water exit via the vent at the top of the vessel. The pumps then take boiler feed water from the bottom of the vessel to the steam generating boiler system.
Tray Type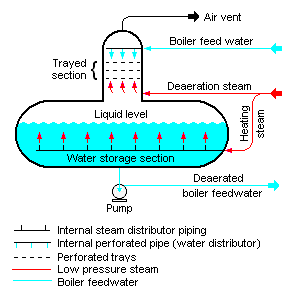
According to STS Canada, the Tray-Type Deaerator is a vertical domed deaeration section mounted on top of a horizontal cylindrical vessel. Boiler feed water enters the vertical deaeration section above the perforated trays and flows downward through the perforations.
Low-pressure deaeration steam enters below the perforated trays and flows upward through the perforations. Some designs use various types of packed bed, rather than perforated trays. This provides good contact and mixing between the steam and the boiler feed water.
The steam strips the dissolved gas from the boiler feed water and exits via the vent valve at the top of the domed section.
The water flows down into the horizontal storage vessel from where it is pumped to the steam generating boiler system. Low-pressure heating steam keeps the stored boiler feed water warm. This enters the horizontal vessel through a sparger pipe in the bottom of the vessel.
DEAERATORS AND SURGE TANKS
Surge tanks can help a deaerator work by averaging out the flow and steam load requirements for the deaerator, and it’s control system. Surge tanks also:
Collects pumped returns
Receives make-up & mixes it with returns
Collects gravity returns
DEAERATOR PROBLEMS?
Watch this video to learn more about Deaerators! Be sure to follow the manufacturers recommendations for maintenance to ensure your boiler system runs as efficiently as possible. If you’re experiencing deaerator problems that have you stumped, reach out for a free estimate or call us at 1-800-237-3141. Our team is here to help!
Bonus: A Great Tool- Required Water And Steam
The department of energy has a DA Calculator that helps you determine the required water and steam flows for your system. Calculation Details:
1: Determine Inlet Water Properties
2: Determine Inlet Steam Properties
3: Determine Feedwater and Vented Steam Properties
4: Determine Feedwater and Vented Mass Flows and Total Outlet Energy Flows
Vented Steam Mass Flow = Vent Rate * Feedwater Mass Flow
Total DA Mass Flow = Vented Steam Mass Flow + Feedwater Mass Flow
Total Outlet Energy Flow = [Feedwater Specific Enthalpy * Feedwater Mass Flow] + [Vented Steam Specific Enthalpy * Vented Steam Mass Flow]
5: Determine Inlet Water and Steam Mass Flows
The inlet mass flows are determined based on an energy and mass flow balance:
Minimum Inlet Energy Flow = Inlet Water Specific Enthalpy * Total DA Mass Flow
Additional Energy Flow Needed = Total Outlet Energy Flow – Minimum Inlet Energy Flow
Inlet Steam Mass Flow = Additional Energy Flow Needed / (Inlet Steam Specific Enthalpy – Inlet Water Specific Enthalpy)
Inlet Water Mass Flow = Total DA Mass Flow – Inlet Water Mass Flow
Assumptions
The Deaerator (DA) Vent Rate is a percent of feedwater flow.
Feedwater leaves the DA as a saturated liquid at DA pressure.
The DA vented gases are assumed to be saturated gas at DA pressure.
The DA has no additional heat or masses losses.
We install commercial and industrial boiler systems for facilities throughout the Midwest and Colorado, serving cities such as Denver, Castle Rock, Boulder, Golden, Westminster, Centennial, Littleton, Omaha, Lincoln, Council Bluffs, Des Moines, Ames, Creston, Storm Lake, Sioux Center, North Platte, Grand Island, Fremont, Hastings, Kearney, Sioux Falls, Rapid City, Vermillion, Brookings, and Yankton.




